Improve
Providing safe, effective, cost-efficient products
We constantly revisit every area of our operations to see what more we can do to improve patient outcomes and optimize cost-efficiency.
Providing the biological drugs Canadians need
Canadian Blood Services is responsible for purchasing and managing the distribution of plasma protein products (PPPs) across Canada. We manage a formulary of about 45 brands on behalf of all provincial and territorial governments except Quebec and distribute these essential medicines to Canadian hospitals. Together, the country’s health systems invest over $750 million annually to provide these essential drugs to patients with hemophilia and other bleeding disorders. Other recipients include people with inherited and acquired immune conditions, as well as burn and trauma victims.
Demand for some PPPs is growing rapidly, as are the complexities of managing the supply of some products. In 2017–2018, the Canadian Blood Services team of medical, financial and supply chain experts continued to collaborate closely with patient groups, prescribing physicians, ministries of health, hospitals and health authorities. Our collective goal is clear: to ensure patients receive consistent, appropriate care while balancing changing utilization patterns against fiscal realities.
Immune Globulin Utilization Growth
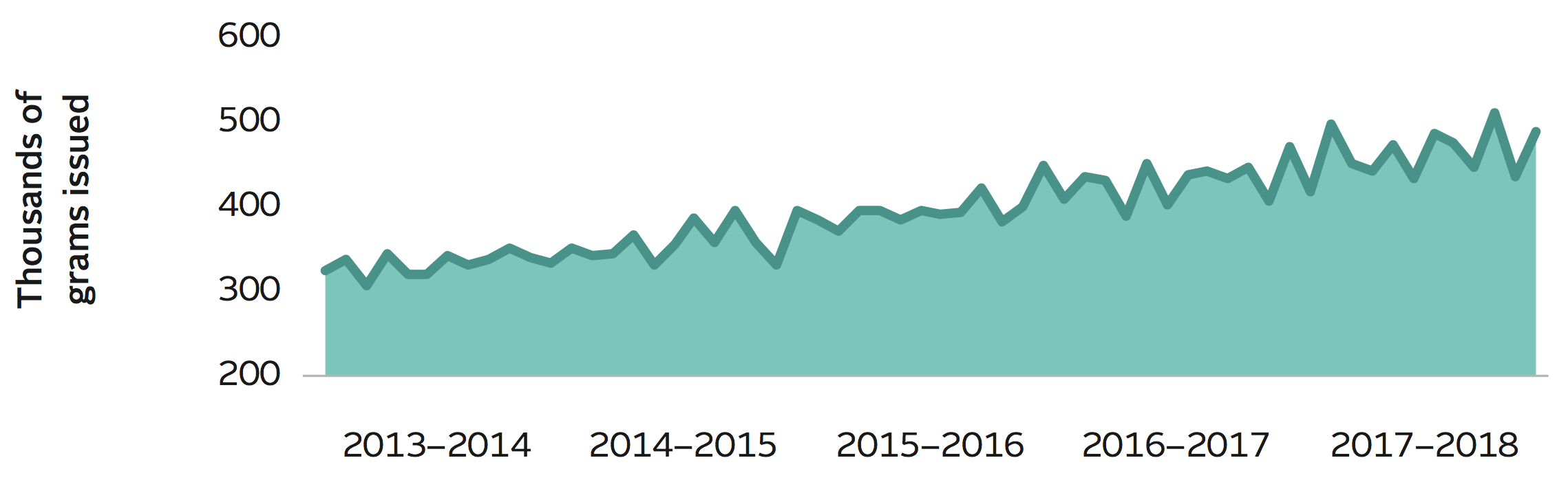
In the past year we once again leveraged our market clout, as a major bulk purchaser of biological drugs, to negotiate favourable contracts with suppliers of PPPs. In the coming yearswe’ll continue working with stakeholders and our government and health-system partners to gain deeper insights into utilization patterns for PPPs, sharpening our collective capacity to anticipate and meet demand.
Extending the shelf life of platelets
During storage in our facilities and at hospitals, platelets must be constantly subject to gentle agitation to prevent them from clumping together and becoming ineffective in medical treatment. They must also be stored at room temperature, making them susceptible to bacterial growth, which in turn could be harmful or even life-threatening to patients receiving transfusions. Our maximum shelf life for platelets has therefore traditionally been five days.
The challenge of how we might make platelets last longer was taken up by Canadian Blood Services researchers working in our Centre for Innovation. A new bacteria-testing process now allows us to extend shelf life by two more days. The new process makes it easier to manage platelet inventories and reduces the number of products that expire before they can be used.
When the solution was introduced during 2017–2018, the number of discarded platelet units went down at our facilities, and hospital re-orders dropped. Demand for platelets decreased, which enabled us to reduce the number of platelet units we collect through apheresis by 5,000 collections. We have begun to see some savings as a result, and we expect to realize even greater savings through the coming year and beyond.
Limiting CMV testing
CMV is a genus of viruses in the herpes family associated with illnesses such as mononucleosis and pneumonia. The virus resides in white blood cells, which Canadian Blood Services removes from whole blood using leukoreduction, a process that is part of how we manufacture red blood cells, plasma and platelets.
Many studies have shown that the residual risk of CMV in leukoreduced blood components is virtually nil. In October 2017, after performing a risk-based decision-making analysis that considered relevant research, Canadian Blood Services implemented a new CMV testing approach, bringing our practices in line with expert recommendations from the National Advisory Committee on Blood and Blood Products. We stopped testing donor blood for anti-CMV antibodies except for a small inventory of blood components that are tested for the sole purpose of intrauterine transfusion. This change will not have an impact on patient safety and will save Canadian health systems an estimated $600,000 a year.



